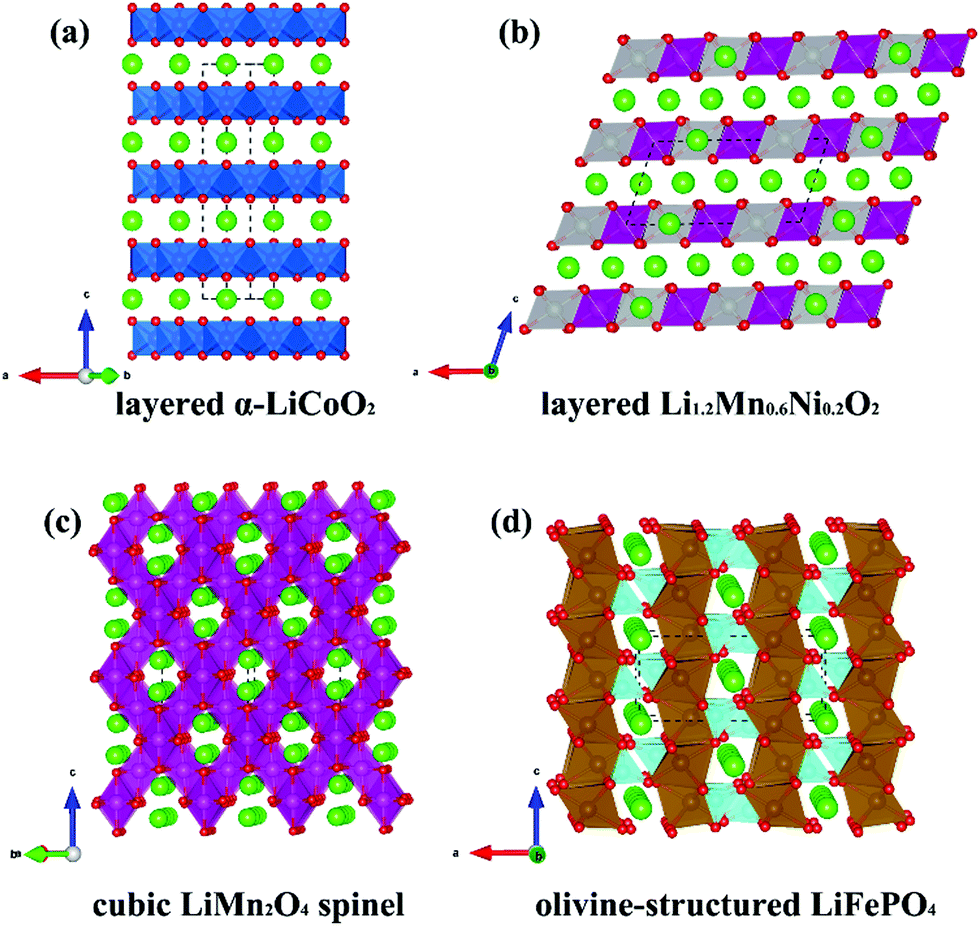
The battery voltage is determined by the potential difference between the cathode and the anode. The higher the Li content, the larger the battery capacity. The additive increases the conductivity of the cathode, and the binder helps maintain the cathode’s structure and maintain good contact with the aluminum foil.ĭifferent cathode materials contain varying amounts of Li. The active material is the source of the Li-ions. (Image: Joule)Ī Li-ion cathode is built on a thin aluminum foil current collector that holds the frame of the cathode coated with a combination of active material, conductive additive, and binder. The two most common are LiCoO2 and LiMn2O4.įigure 1: Initially, various Li-free cathodes were paired with metallic Li anodes to produce rechargeable batteries the development of Li-ion batteries required the invention of various Li compounds to provide the Li ions for intercalation/deintercalation in a graphite anode. A range of cathode materials has been developed that deliver specific levels of performance, including Li2MnO3, LiFePO4, Li2MO3-LiM’O2, Li(NiMnCo)1/3O2 (also called NMC) (Figure 1). The cathode is the source of the ions inserted into the lattice structure of the anode during charging (intercalation) and extracted from the anode during discharge (deintercalation). Li-ion batteries are based on the intercalation and deintercalation of Li ions. 
The anodes in Li-ions are made from materials such as graphite, silicon, and silicon oxides that can act as hosts for Li-ions delivered by the cathode (a discussion of Li-ion anodes can be found in “Li-ion Parts 3 – Anodes”) Today’s Li-ions were developed to eliminate elemental Li by moving the active material from the anode to the cathode, resulting in a much safer structure. However, these Li-metal-based rechargeable batteries were inherently unsafe due to Li dendrites piercing the separator, shorting the battery, and causing fires. From the 1960s to the early 1980s, various Li-free cathodes were paired with metallic Li anodes to produce rechargeable batteries. Today’s Li-based cathode materials are a relatively new development. This FAQ begins with a brief look at the longer-term trajectory of Li-ion cathode developments, dives into cathode structures and chemistries currently being used, and closes by reviewing efforts to develop Co-free, and Li-free cathodes, including the use of disordered rocksalts, needed to support long-range electric vehicles, electric aircraft, and other advanced applications. Today’s common cobalt (Co) and manganese (Mn) based cathodes were developed to overcome safety concerns with Li-metal anodes. Among the various components involved in a lithium-ion cell, cathodes (the positive or oxidizing electrodes) currently limit the energy density and dominate the battery cost.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
